Pin
Pin Paul Conyngham with his dog Rosie / Photo courtesy of University of New South Wales
Synopsis: When Rosie, an eight-year-old rescue dog in Sydney, was diagnosed with deadly mast cell cancer, her owner Paul Conyngham refused to give up. A tech entrepreneur with 17 years in machine learning, he turned to ChatGPT and AlphaFold to decode her tumor’s DNA. Working with UNSW scientists, he built a personalized mRNA vaccine — the first of its kind ever made for a dog. Her tumor shrank by 75%. Now the world is paying attention.
Rosie is an eight-year-old rescue dog — a Staffordshire Bull Terrier-Shar Pei cross — adopted by Sydney-based tech entrepreneur Paul Conyngham in 2019. She was scrappy, loyal, and apparently had a thing for chasing rabbits. She was also, by all accounts, Paul’s whole world.
In 2024, Rosie was diagnosed with a deadly mast cell cancer. This is the most common form of skin cancer in dogs, and once it spreads, conventional medicine doesn’t have great answers for it. Conyngham tried everything conventional veterinary medicine offered — multiple surgeries, chemotherapy, immunotherapy. The chemo slowed the tumor spread but failed to shrink the growths. Vets estimated Rosie had between one and six months to live.
That six-month clock would terrify most people into grief. Paul Conyngham used it as a deadline. He didn’t have a biology degree, no lab coat, no pharmaceutical connections. What he had was data skills, an internet connection, and the stubborn conviction that the answer had to be somewhere in the numbers.
Table of Contents
The Man Behind the Mission
 Pin
Pin Paul Conyngham with his dog / Photo courtesy Paul Conyngham
Paul Conyngham is a data scientist and machine learning engineer with 17 years in the field — but no background in biology. He co-founded a Sydney consultancy called Core Intelligence Technologies and had served as a director for the Data Science and AI Association of Australia. In other words, he knew how to work with complex data. He just hadn’t pointed that skill at medicine before.
When conventional veterinary medicine failed Rosie, Conyngham viewed her cancer not as a biological dead end, but as a data problem. That mental shift is actually the most important part of this story. He wasn’t trying to play doctor. He was trying to find the right question to ask — and then use the right tools to answer it.
His background made him uniquely suited for this. Most pet owners would have accepted the prognosis or exhausted the standard treatment options. Conyngham looked at gigabytes of genetic data and saw a puzzle waiting to be solved. What followed would stun even the scientists who helped him.
ChatGPT as the First Step
 Pin
Pin Paul Conyngham with his dog / Photo courtesy Paul Conyngham
Conyngham turned to ChatGPT and came up with a plan on how to approach the problem. That sentence sounds casual, but it represents something genuinely new — a person with no medical training using an AI chatbot to navigate one of oncology’s most complex frontiers: personalized cancer immunotherapy.
OpenAI’s ChatGPT suggested immunotherapy and directed Conyngham to the University of New South Wales Ramaciotti Centre for Genomics. He followed that lead. Based on the AI chatbot’s recommendation, he reached out to the Ramaciotti Centre and paid $3,000 to have Rosie’s DNA sequenced.
For context, DNA sequencing used to cost millions of dollars and take years. Today, $3,000 can map an entire tumor genome in weeks. That falling cost is part of what made Conyngham’s plan even possible. He was also using ChatGPT not as a magic answer machine, but as a research librarian — asking it where to look, who to contact, and what questions to pursue. The AI didn’t design the vaccine. It helped him figure out where to start.
Key AI tools Conyngham used:
- ChatGPT — research planning, identifying scientists, interpreting scientific literature
- AlphaFold (Google DeepMind) — modeling 3D protein structures from tumor DNA
- Genomic sequencing — mapping Rosie’s healthy and tumor DNA to find cancer-driving mutations
Decoding the Tumor's DNA
 Pin
Pin Photo courtesy Uncover.ai
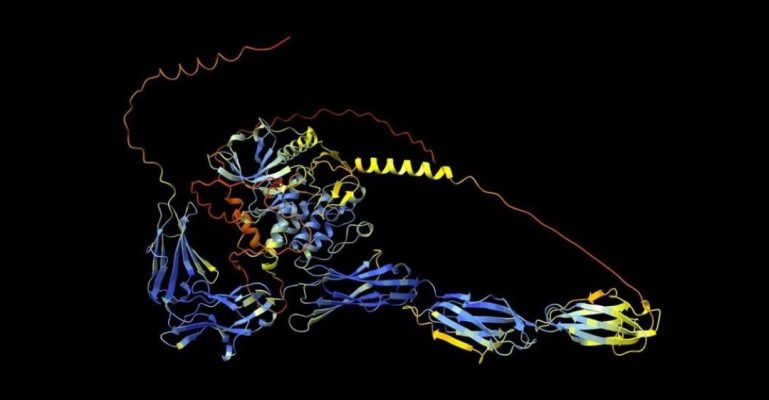
Once Conyngham had Rosie’s genomic data in hand, the real technical work began. He used AlphaFold, an AI program developed by Google DeepMind that predicts the 3D structure of proteins, to model a protein called c-KIT — which drives mast cell cancer in dogs. Understanding how that protein was mutated was essential to figuring out how to target it.
From those models, he identified which mutated proteins — known as neoantigens — were most likely to trigger a meaningful immune response. Neoantigens are essentially flags on the surface of cancer cells that the immune system can learn to recognize and attack. Finding the right ones is like identifying the serial number on a criminal — specific, unmistakable, and actionable.
ChatGPT helped interpret complex scientific metrics, and this data-driven process identified 10 to 20 high-priority target peptides. These would become the blueprint for Rosie’s vaccine. What Conyngham produced was condensed into half a page of formulas describing an mRNA sequence — compact, precise, and ready to hand to a lab.
The Scientists Who Said Yes
 Pin
Pin Martin Smith / Photo courtesy UNSW
At this point in the story, Conyngham had a plan on paper. What he needed was a team willing to make it real. Martin Smith, an associate professor of computational biology and director of the Ramaciotti Centre for Genomics at UNSW, recalled his reaction when Conyngham presented his analysis: “Paul was relentless. He called and told me he had analyzed the data, found mutations of interest, used AlphaFold to find the mutated proteins, identified potential targets, and matched them to drugs. I’m like, ‘Woah, that’s crazy!'”
That reaction — somewhere between amazement and disbelief — tells you something important. Even professional researchers hadn’t seen a layperson pull off this kind of analysis before. While an immunotherapy treatment that looked like a good fit for Rosie was identified, the drugmaker wouldn’t provide it. A closed door. Most people would have stopped there.
Smith then introduced Conyngham to Pall Thordarson, director of the UNSW RNA Institute. Thordarson used Conyngham’s data, condensed into a half-page formula, to create a custom mRNA vaccine for Rosie. It took less than two months. The collaboration between a determined dog owner and some of Australia’s top scientists had produced something the world had never seen before.
What Is an mRNA Cancer Vaccine, Anyway?
Most people got their first real introduction to mRNA technology during the COVID-19 pandemic. But the science behind it has been quietly building toward cancer treatment for years. To create personalized cancer vaccines, scientists genetically sequence DNA from a patient’s tumor, looking for mutated proteins known as neoantigens. Machine learning algorithms select antigens most likely to provoke an immune response. They then create mRNA blueprints that encode instructions for making those neoantigens. These are packaged into lipid nanoparticles — fat bubbles — for injection, using the same delivery technology as the Pfizer/BioNTech and Moderna COVID vaccines.
In plain terms: the vaccine teaches your immune system to recognize the specific fingerprints of ‘your’ cancer and attack them. It’s not a generic treatment. It’s built from your own tumor’s DNA, tailored entirely to you — or in Rosie’s case, to her.
Nanomedicine pioneer Pall Thordarson, director of UNSW’s RNA Institute, described what Rosie’s case demonstrates: “This is still at the frontier of where cancer immunotherapeutics are — and ultimately, we’re going to use this for helping humans. What Rosie is teaching us is that personalized medicine can be very effective, and done in a time-sensitive manner, with mRNA technology.”
The Injection and the Wait
The bespoke mRNA vaccine was encased in lipid nanoparticles by Professor Thordarson’s lab and administered to Rosie over the Christmas break in December 2025 at the University of Queensland. After months of data work, lab collaboration, and navigating regulatory hurdles around experimental veterinary treatments, it came down to a single injection.
One week after the initial injection, the tumor began to visibly shrink. That timeline is remarkable in cancer treatment terms. Immunotherapy responses can take weeks or months to appear. Seeing movement within a week caught even the treating team off guard.
By January 2026, Rosie had regained enough mobility to jump a fence at the dog park after spotting a rabbit. For anyone who has watched a sick animal lose the will to move, that image says everything. Conyngham described December as the month Rosie “started to shut down and be a bit sad.” Six weeks later, she was airborne over a fence. That’s not a statistic. That’s a life returning.
The Results That Stunned Scientists
The tumor on Rosie’s leg shrank by 75%. That’s not a minor improvement — in advanced mast cell cancer that had already resisted chemotherapy and surgery, a 75% reduction is the kind of number that makes researchers stop mid-sentence.
Rachel Allavena, the professor who administered the vaccine, confirmed the results: “Rosie’s cancer was really, really advanced, but one tumor has shrunk quite a lot, probably by half. Even her coat looks glossier, and she just seems much happier and healthier.”
Martin Smith put it more bluntly: “It was like, holy crap, it worked!” He added, “It raises the question — if we can do this for a dog, why aren’t we rolling this out to all humans with cancer?” That question is now echoing through research institutions and media outlets around the world. Not all of Rosie’s tumors responded equally — Conyngham is under no illusion that this is a cure, but he believes the treatment has bought Rosie significantly more time and quality of life. He’s already working on a second vaccine targeting the tumor that didn’t respond.
The Bigger Picture
Rosie’s story didn’t happen in a vacuum. What a tech entrepreneur with a sick dog and a ChatGPT subscription demonstrated in a matter of months — going from tumor biopsy to custom mRNA treatment using AI — is the same process that Moderna, Merck, and BioNTech are spending billions to industrialize for human patients.
The gap between Conyngham’s scrappy citizen-science project and those billion-dollar pipelines is largely regulatory and logistical — not scientific. Rosie’s treatment was possible because veterinary experimental treatments face lighter regulatory scrutiny than human medicine. Scaling this to a standardized treatment — for dogs or for humans — would require years of regulatory work that this single case doesn’t shortcut.
Conyngham called the experience an example of citizen science, and researchers think it could help shape the future of cancer treatment for both animals and people. “There’s actually a chance that for some cancers, we can change it from being a terminal sentence to a manageable disease,” he said. That’s not hype from a tech bro. That’s a data engineer who just watched it happen, speaking carefully, with evidence behind him.
The Cost Question: Can Anyone Do This?
One of the first questions people ask after hearing Rosie’s story is: ‘could I do this for my dog? For myself?’ The honest answer right now is complicated.
Conyngham spent \$3,000 to sequence Rosie’s DNA, but without interested scientists volunteering their time and resources, production of mRNA nanoparticles would be far more costly. One estimate puts the price tag at $100,000 per person. Getting new cancer treatments through clinical trials can cost hundreds of millions of dollars.
Conyngham was uniquely positioned — he had the technical skills to read genomic data, the professional network to reach elite scientists, and the persistence to keep pushing when doors closed. Most people don’t have those advantages. Over time, with evolving AI and automation, companies may be able to simplify and speed up the process and bring costs down.
That’s the trajectory this story points toward: not a solution available to everyone today, but a glimpse of where medicine is headed — faster, more personal, and eventually more accessible than the current system allows.
Rosie, Rabbits, and What Comes Next
After Rosie’s story went viral, Conyngham posted a Google Form for people who want to explore the same kind of vaccine for their dogs, saying, “It would be my dream to make this process something everyone could have access to.” That’s a long road, but it’s being walked.
Thordarson has stated that this case demonstrates how technology can “democratize” the process of designing cancer vaccines. The tools Conyngham used — ChatGPT, AlphaFold, genomic sequencing — are all available right now. What’s still rare is the specific combination of technical skill, scientific access, and sheer stubbornness that Conyngham brought to the problem.
But here’s the thing about breakthroughs: they tend to start with one case. One dog, one owner, one team of scientists who said yes. Rosie received her vaccine over Christmas 2025. Her coat is glossier. She’s jumping fences. And somewhere at UNSW, researchers are asking why this can’t be done for every cancer patient who needs it. The answer, hopefully, is that it can — and Rosie just showed everyone the first draft of how.
FAQs
Not fully. Most tumors shrank by up to 75%, but one didn’t respond. Conyngham is developing a second vaccine targeting that tumor. Researchers call the results remarkable, not a cure.
Conyngham spent $3,000 on DNA sequencing. Full production would cost far more without volunteer scientists — estimates suggest $100,000+ per patient at current scale.
Not yet as a standard treatment. Conyngham opened a Google Form for interested pet owners and hopes to make the process more accessible in the future.
ChatGPT for research planning and literature guidance, and AlphaFold by Google DeepMind for modeling the 3D structure of Rosie’s mutated tumor proteins.
Scientists say yes — in principle. The same mRNA approach is already in human trials at Moderna and Merck. Rosie’s case proves the science can work fast and personally. Regulatory approval is the longer road.