 Pin
Pin Photo courtesy of Horizon Dwellers
Synopsis: A vaccine works by showing your immune system what a threat looks like — without the actual danger. Traditionally, making one took 10 to 15 years. Today, AI is compressing that timeline dramatically. From identifying the right target protein to designing custom mRNA sequences, artificial intelligence is now embedded in every stage of vaccine development. This article breaks down exactly how a vaccine is made and where AI steps into each part of the process.
Most people know vaccines prevent disease, but fewer understand the elegant mechanism behind that protection. Your immune system is essentially a biological memory bank. Every time it encounters a new threat — a virus, a bacteria, a rogue cancer cell — it mounts a defense and, crucially, remembers how it did it. The next time that same threat appears, the response is faster and stronger.
A vaccine is simply a shortcut to that memory. Instead of letting the real disease teach your immune system the hard way, a vaccine introduces a harmless preview — a fragment of the pathogen, a weakened version of it, or in the case of modern mRNA vaccines, just the instructions to make one recognizable piece of it. Your immune system reads the preview, mounts a practice defense, and stores the memory. When the real thing arrives, it’s already prepared.
Understanding how a vaccine is made using AI starts here — with this core mission. Everything that follows in the manufacturing process is in service of one goal: finding the best possible preview to show the immune system, and delivering it safely.
- The immune system has two layers: innate (fast, general response) and adaptive (slow, specific, memory-forming)
- Vaccines primarily train the adaptive immune system
- The piece of the pathogen a vaccine uses is called an “antigen”
- Choosing the right antigen is the single most critical decision in vaccine design
Table of Contents
Step 1: Identifying the Enemy (Antigen Discovery)
 Pin
Pin Photo courtesy of Horizon Dwellers
Before anything can be made, scientists need to know what to target. This is antigen discovery — the process of studying a disease-causing agent and finding the one specific protein or molecule that the immune system can reliably recognize and attack. It sounds straightforward, but in practice it’s like searching for a single useful address in an entire city’s phone directory.
A virus might have dozens of surface proteins. A cancer cell might carry hundreds of mutations. Not all of them make good vaccine targets — some are too similar to healthy tissue (risking autoimmune responses), some are hidden from immune detection, and some simply don’t trigger a strong enough reaction to matter. Scientists need the ones that are visible, distinct, and immunologically powerful.
Traditionally, this discovery process involved years of laboratory experiments — growing pathogens in cell cultures, testing proteins one by one, observing immune responses in animals. It was painstaking, slow, and expensive. A wrong turn early could waste years. The entire HIV vaccine effort has been stalled partly on this problem for four decades — not for lack of trying, but because finding the right antigen in a highly mutating virus is extraordinarily difficult.
How AI Transforms Antigen Discovery
 Pin
Pin Photo courtesy of Horizon Dwellers
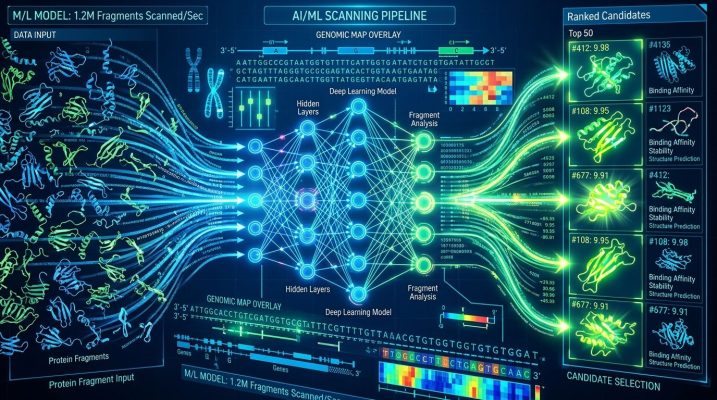
This is where AI enters the story and immediately changes the math. Instead of testing antigens one by one in a lab, machine learning models can analyze entire genomic datasets and predict which protein fragments are most likely to trigger a strong immune response — before a single experiment is run.
These models are trained on vast libraries of known antigens: proteins that have already been proven to work in existing vaccines or natural immunity. They learn the patterns — the structural features, the amino acid sequences, the binding characteristics — that make an antigen effective. Then, given a new pathogen or a tumor’s mutated proteins, the AI ranks candidates by predicted effectiveness in hours.
Paul Conyngham used exactly this approach for Rosie’s cancer vaccine. He fed her tumor’s genomic data into AI tools, which identified the mutated proteins — called neoantigens — most likely to provoke her immune system into attacking the cancer. What might have taken a team of researchers months of wet-lab work was condensed into a data analysis task measured in days.
- Traditional antigen discovery: months to years, experimental, costly
- AI-assisted antigen discovery: hours to days, predictive, scalable
- Key AI tools used: machine learning classifiers, neural networks trained on immunogenicity data
- Output: a ranked shortlist of antigen candidates ready for the next stage
Step 2: Understanding the Protein's Shape (AlphaFold)
 Pin
Pin Photo courtesy of Horizon Dwellers
Once you’ve identified a candidate antigen, the next challenge is understanding its three-dimensional structure. This matters enormously because a protein’s shape determines everything — how the immune system grabs onto it, whether antibodies can bind to it effectively, and whether it’s stable enough to survive inside a vaccine formulation.
For 50 years, determining a protein’s 3D structure required a laborious technique called X-ray crystallography. You had to grow the protein into a crystal, fire X-rays at it, and interpret the diffraction patterns — a process that could take years per protein and required highly specialized equipment and expertise. Many proteins resisted crystallization entirely and remained structurally unknown.
In 2020, Google DeepMind’s AlphaFold changed this permanently. AlphaFold is an AI model trained on every known protein structure in existence. Given just the genetic sequence of a protein, it predicts the full 3D folded structure with remarkable accuracy — in minutes. When Conyngham identified the c-KIT mutation driving Rosie’s mast cell cancer, he used AlphaFold to visualize exactly how that mutated protein was shaped, and therefore how an immune response might physically interact with it. A task that would have required a structural biology lab and months of work was completed on a laptop.
Step 3: Choosing the Vaccine Type
 Pin
Pin Photo courtesy of Horizon Dwellers
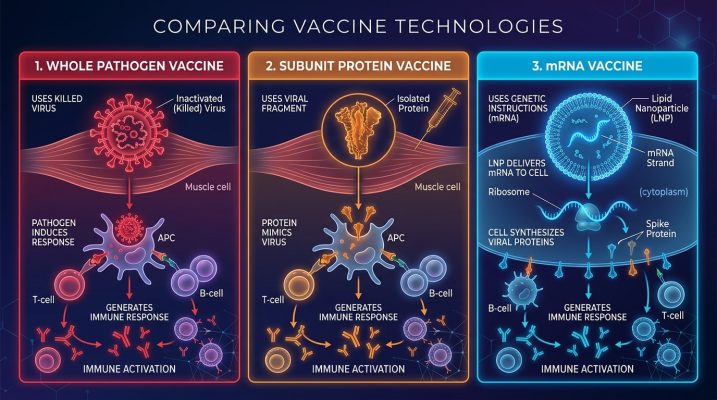
With a confirmed antigen and an understanding of its structure, scientists choose the delivery strategy — what form the vaccine will take. There are several fundamentally different approaches, and each has its own strengths depending on the disease.
The oldest approach uses weakened or killed versions of the whole pathogen. The polio vaccine and the traditional flu shot work this way. They’re effective but slow to manufacture because you need to grow the actual pathogen in large quantities, which is inherently risky and time-consuming.
Subunit vaccines take just one piece of the pathogen — usually a surface protein — and use that alone. The hepatitis B vaccine works this way. Cleaner and safer to produce, but often requires chemical additives called adjuvants to boost the immune response since you’re showing the immune system such a small piece of the threat.
The newest approach is mRNA vaccines, which Rosie’s cancer vaccine used. Instead of delivering the antigen itself, you deliver the genetic instructions to make it. Your own cells temporarily produce the antigen, your immune system responds and forms memory, and then the instructions degrade and vanish. Nothing foreign stays in the body. This approach is fast to design, highly adaptable, and precisely personalizable — which is why AI-assisted cancer vaccines almost universally use it.
Step 4: Writing the mRNA Sequence
 Pin
Pin Photo courtesy of Horizon Dwellers
Once you know which antigen to target and what it looks like in 3D, you need to write the mRNA — the actual molecular instruction code that will tell your cells to make it. This is essentially programming at the biological level, and it’s more nuanced than it first appears.
An mRNA strand is a sequence of four chemical letters — A, U, G, and C — arranged in a specific order that your cells’ ribosomes can read and translate into a protein. Writing the right sequence means encoding not just the protein itself, but also regulatory signals that control how efficiently it’s read, how stable the mRNA is inside the cell, and how long the protein is produced before the instructions degrade.
AI tools now assist with every one of these decisions. Given a target antigen sequence, algorithms can optimize the mRNA code — choosing synonymous codons (different letter combinations that encode the same amino acid) that are more efficiently read by human cells, adding stabilizing chemical modifications, and predicting the secondary structure the mRNA will fold into. Moderna and BioNTech both use proprietary AI systems for this optimization. Conyngham’s team used computational tools to condense Rosie’s entire vaccine blueprint into half a page of formulas — precise, optimized, and ready for manufacturing.
Step 5: Packaging in Lipid Nanoparticles
 Pin
Pin Photo courtesy of Horizon Dwellers
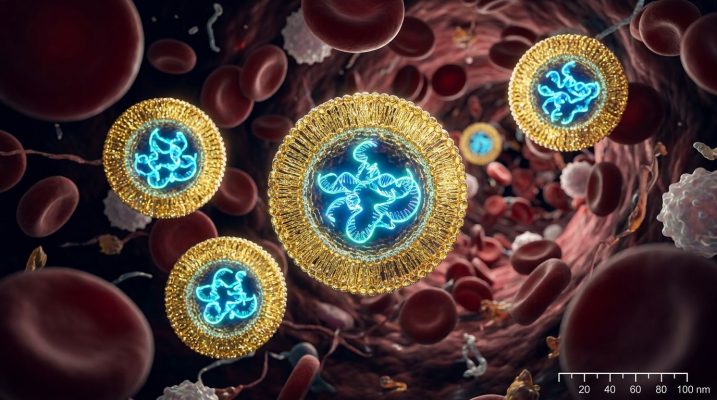
Now the mRNA sequence exists on paper — or rather, in a computer. Before it can be injected, it needs a delivery vehicle that can protect it during transit through the bloodstream and get it safely inside a cell. This is where lipid nanoparticles come in.
Lipid nanoparticles are microscopic spheres made of fat molecules, roughly 80 to 100 nanometers across — small enough that 800 of them could sit side by side across the width of a single human hair. Their fat layer mimics the natural membrane of human cells, which allows them to merge with cells and release their cargo inside. Without this packaging, injected mRNA would be destroyed by enzymes in the bloodstream within minutes.
Manufacturing lipid nanoparticles at laboratory scale is one thing — it involves carefully mixing specific lipid formulations under precisely controlled conditions so they self-assemble around the mRNA payload. Doing it at industrial scale, consistently and affordably, is a significant engineering challenge. AI is increasingly used here too, helping predict optimal lipid compositions for different mRNA payloads and modeling how nanoparticles will behave in different tissue environments. For Rosie’s vaccine, Professor Pall Thordarson’s lab at UNSW handled this step manually — but the formulation itself was guided by computational chemistry.
Step 6: Testing — From Lab to Living Beings
 Pin
Pin Photo courtesy of Horizon Dwellers
With a formulated vaccine in hand, the next phase is testing — verifying that it actually does what the theory predicts. This stage is traditionally the longest and most expensive part of the entire process, often consuming the majority of the 10–15 year development timeline.
Preclinical testing comes first: cell cultures, then animal models. Scientists check whether the immune system responds at all, whether the response is the right kind (antibodies, T-cells, or both), whether the vaccine causes harm, and what dose range is safe. Then come human clinical trials in three phases — Phase 1 checks safety in a small group, Phase 2 expands to hundreds of people checking efficacy and side effects, and Phase 3 involves thousands of participants to confirm effectiveness across diverse populations.
AI is transforming this stage in two ways. First, predictive models can simulate immune responses computationally before any animal or human testing — identifying likely failures early and avoiding years of dead ends. Second, AI tools are accelerating the analysis of clinical trial data, identifying signals of efficacy or safety concern faster than traditional statistical methods. During COVID vaccine development, AI-assisted data analysis helped compress Phase 3 trial interpretation from months to weeks. For personalized cancer vaccines like Rosie’s, AI-generated predictions about immune response were validated almost immediately when her tumor began shrinking within one week of injection.
Step 7: Manufacturing at Scale
 Pin
Pin Photo courtesy of Horizon Dwellers
Passing clinical trials is one achievement. Making millions of doses reliably, cheaply, and quickly is an entirely different challenge — and historically, it’s where vaccine rollouts have stalled. The COVID-19 pandemic exposed just how fragile global vaccine manufacturing capacity actually was.
mRNA vaccines have an advantage here: the manufacturing process is largely the same regardless of what mRNA sequence you’re producing. Once you have the infrastructure to make lipid nanoparticles and synthesize mRNA, switching to a different vaccine target requires changing the sequence — not rebuilding the factory. This is fundamentally different from older vaccine platforms that required growing specific pathogens under biosafety conditions.
AI is embedded throughout modern vaccine manufacturing. Process optimization algorithms monitor bioreactor conditions in real time, catching deviations before they become batch failures. Quality control AI analyzes spectroscopic data from production runs to detect impurities faster than human inspectors. Supply chain models predict raw material demand and flag potential shortages weeks in advance. For personalized cancer vaccines — where each batch is custom-made for one patient — AI is what makes the economics even conceivable. Automated pipelines that sequence a tumor, design the vaccine, and queue manufacturing with minimal human intervention are already being built by companies like Moderna and BioNTech.
The Full AI-Powered Pipeline Today
 Pin
Pin Photo courtesy of Horizon Dwellers
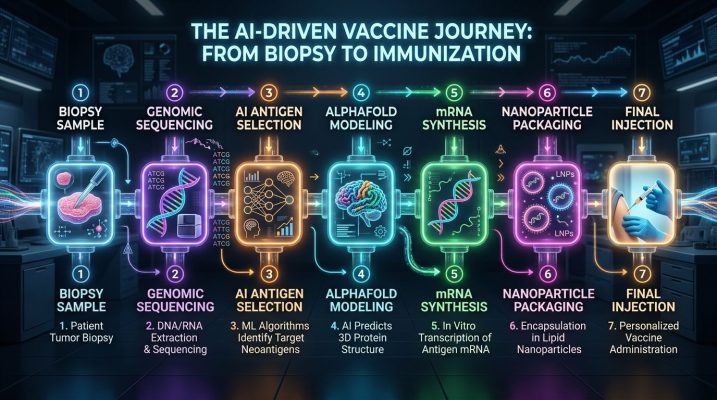
Pull all the steps together and you get a picture of how dramatically the vaccine development landscape has shifted. What once required a decade of sequential experiments — each waiting for the last to finish — can now run in parallel, with AI accelerating analysis at every node.
The modern AI-assisted vaccine pipeline flows like this: a biopsy or blood sample provides biological material, which is sequenced to map the relevant DNA. AI scans that genomic data and surfaces the top antigen candidates within hours. AlphaFold models their 3D structures. An mRNA sequence is computationally optimized. Lipid nanoparticles are formulated using AI-guided chemistry. Preclinical response is predicted in silico before any animal testing begins. Manufacturing is monitored by process AI. And if the first vaccine doesn’t fully work — as happened with one of Rosie’s tumors — the data feeds back into the pipeline for a second-generation design.
This isn’t a distant future scenario. Moderna’s mRNA-4157 personalized cancer vaccine — built on exactly this kind of AI pipeline — is currently in Phase 3 clinical trials for melanoma. BioNTech has a similar program. What Paul Conyngham assembled informally with off-the-shelf tools and UNSW scientists is, in principle, the same architecture that major pharmaceutical companies are now spending billions to industrialize and standardize.
- Timeline comparison: Traditional vaccine development: 10–15 years. AI-assisted mRNA vaccine: 2–5 years for novel targets, months for personalized cancer variants
- Cost trajectory: Personalized cancer vaccines currently cost $100,000+. AI automation is projected to bring this down significantly within a decade
- Key companies: Moderna, BioNTech, Gritstone Bio, Neon Therapeutics, Vaccibody
What This Means for the Future of Medicine
 Pin
Pin Photo courtesy of Horizon Dwellers
The implications of AI-assisted vaccine development reach well beyond cancer or even infectious disease. What’s really happening is that medicine is shifting from a population model to a personal model. Traditional drugs and vaccines are designed for the average patient — they work well for most people, imperfectly for many, and not at all for some. Personalized medicine, powered by AI, is designed for you specifically, based on your genetics, your tumor’s unique fingerprint, your immune system’s particular characteristics.
This shift is still early. Regulatory frameworks weren’t built for personalized biologics. Manufacturing infrastructure is only beginning to scale. Access remains profoundly unequal — Conyngham could pursue this path because he had technical skills, scientific connections, and the resources to sequence Rosie’s genome. Most people don’t have those advantages yet.
But the trajectory is clear. The tools — genomic sequencing, AlphaFold, mRNA synthesis, AI-guided formulation — are all available today. Their costs are falling. Their accessibility is growing. Rosie’s story sits at the visible tip of this transformation, which is part of why it resonated so widely. She wasn’t just a dog who beat cancer. She was a proof of concept that personalized AI-designed medicine works — fast, precisely, and in a living patient. The question the scientific community is now asking is not whether this becomes standard care, but how soon — and for whom first.
This article was researched using publicly available scientific literature, clinical trial data, and verified news reporting. It is intended to educate general readers on how a vaccine is made using AI. It does not promote any specific treatment, product, or medical protocol. Always consult a licensed medical professional before making health-related decisions.
FAQs
AI has compressed early-stage vaccine design from years to weeks. For personalized cancer vaccines, the full process — from tumor biopsy to injection — can now take as little as a few months.
An mRNA vaccine delivers genetic instructions to your cells to make a target protein. Your immune system responds, forms memory, and the mRNA degrades completely. No DNA alteration occurs. Billions of doses have confirmed its safety profile.
Not yet. AI accelerates and guides each step — antigen selection, protein modeling, sequence optimization — but human scientists still validate, formulate, test, and administer the vaccine. AI is the navigator; scientists are the drivers.
AlphaFold is Google DeepMind’s AI that predicts a protein’s 3D shape from its genetic sequence in minutes. Knowing a protein’s shape is essential for designing vaccines that the immune system can effectively target.
Currently they cost $100,000+. But as AI automation scales and manufacturing standardizes, costs are expected to fall significantly — similar to how genomic sequencing dropped from millions to thousands of dollars in a decade.































